The Emergence of Antibody-Drug Conjugates as a Core Oncology Modalit
Antibody-drug conjugates (ADCs) have moved from a niche therapeutic concept to a validated and increasingly important oncology modality. The market has expanded rapidly in recent years, supported by strong clinical adoption, continued regulatory approvals, and growing investment from both large pharmaceutical companies and emerging biotech players.
ADCs are a class of targeted therapeutics that combine chemotherapy with targeted delivery to treat certain blood cancers and solid tumors. The approach selectively targets cancerous cells, administering small, highly potent doses of chemotherapy directly into the cells. Compared to conventional cancer therapies, ADCs destroy malignant cells while reducing systemic toxicity and impact on healthy tissue. Since the first regulatory approval in 2000, advances in linker chemistry, payload design, and antibody engineering have improved stability, targeting precision, and overall therapeutic performance.
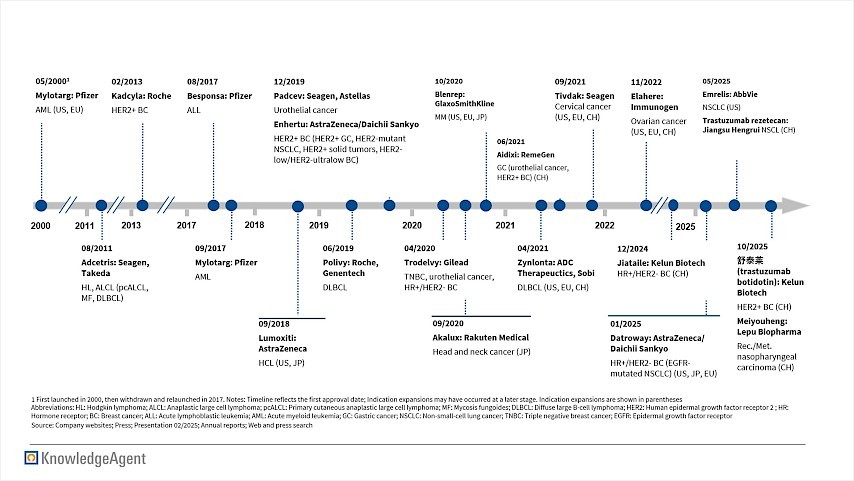
This technological progress has been accompanied by market expansion. Between 2020 and 2024 alone, the global ADC market grew at a CAGR of 36 percent, reflecting both rising clinical adoption and an expanding pipeline. As of early 2026, more than twenty ADCs are commercially available, supported by over a dozen FDA approvals across solid tumors and hematologic malignancies, as illustrated in Figure 1.
Figure 1: ADC Approvals Timeline
The ADC Commercial Landscape Remains Concentrated
Strategic activity within the sector reflects this transition toward a more established market. Large-scale transactions such as Pfizer’s acquisition of Seagen in 2023 and AbbVie’s acquisition of ImmunoGen in 2024 indicate increasing consolidation and commitment from major pharmacos. The competitive landscape is characterized by a concentration of commercial revenues among a limited number of dominant players, alongside increasing innovation from smaller and emerging players. Pfizer, Roche and the AstraZeneca and Daiichi Sankyo have established a strong market presence, reflected in commercial success and blockbuster products.
Pipeline Momentum Is Driven by Emerging Players From China
eyond these leading incumbents, development momentum is increasingly driven by Chinese biopharmaceutical companies. This activity is characterized by a focus on speed, cost efficiency, and parallel development approaches. Kelun Biotech, Jiangsu Hengrui, and Lepu Biopharma are advancing multiple ADC candidates globally, and their launch activity has gained traction in late 2025.
Kelun Biotech launched Jiataile in December 2024 and Trastuzumab Botidotin in October 2025. It is closely followed by Jiangsu Hengrui and Lepu, which also recorded successful launches in China in 2025. At the same time, smaller Western innovators such as Tubulis are addressing bottlenecks in linker stability, conjugation precision, and manufacturability. Together, these dynamics point to a more fragmented and competitive innovation environment, even as commercial revenues remain concentrated. The player landscape remains full (see Figure 2), with earlier phase players starting to explore ADCs in indications beyond oncology, including for example in the autoimmune, neurology or fibrotic diseases fields.
Figure 2: ADC Competitive Landscape by Clinical Phase

ADC Market Forecast to 2034: Scenario Analysis
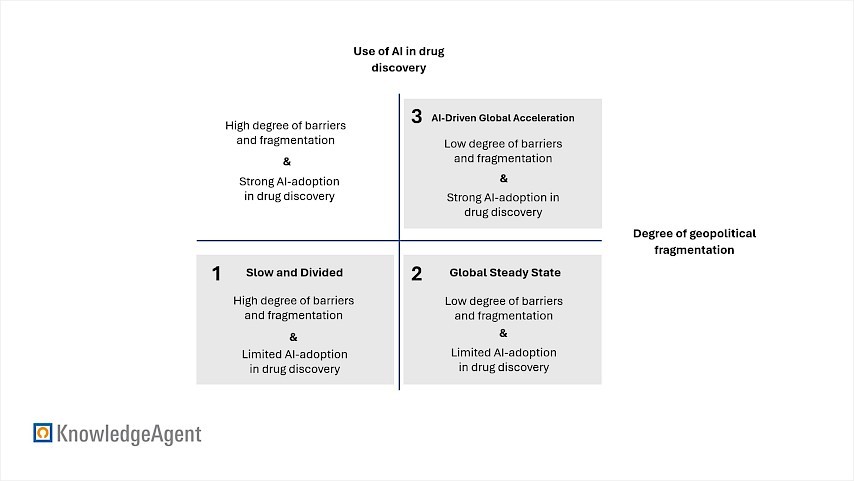
Against the backdrop of this broad pipeline, players can ask themselves how the competitive landscape will evolve. Many market analyses primarily focus on the pipeline and R&D side of the market. In our approach, we wanted to reflect the entire range of factors shaping the ADC market. Thus, we chose a scenario-based approach which combines bottom-up market sizing with a scenario-based framework extending to 2034. This approach is based on two main critical uncertainties: the degree of geopolitical fragmentation as well as the use of AI in drug discovery. In addition, payer acceptance and reimbursement act as a cross-scenario driver. An overview of the scenario axes and uncertainties is shown in Figure 3.
Figure 3: Scenario Axes and Critical Uncertainties
Our modeling projects three distinct trajectories through 2034. In our conservative "Slow and Divided" scenario, geopolitical fragmentation disrupts supply chains and constrains capacity. Yet, even with these headwinds, strong clinical demand drives the market to USD 40.9 bn, representing a CAGR of 11.6%. In our baseline "Global Steady State" scenario, fundamentals such as aging demographics and pipeline maturity propel the market to USD 45.1 bn at a 12.7% CAGR. Finally, the optimistic "AI-Driven Global Acceleration" scenario assumes that artificial intelligence successfully drives drug discovery outcomes, while the geopolitical outlook remains stable. This outlook yields to a market of USD 48.6 bn at a 13.6% CAGR, highlighting the potential value unlock from AI-driven drug discovery methods. An overview of the three market forecasts across the scenarios is provided in figure 4.
Figure 4: Market Forecast
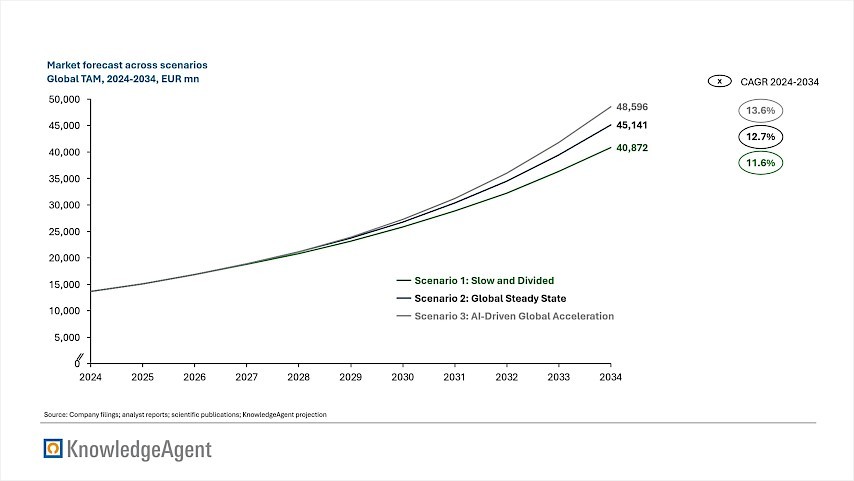
The graph shows that, across scenarios, differences in projected outcomes remain relatively limited, indicating that structural demand drivers are a primary determinant of market growth. Variations in trajectory are primarily linked to the extent to which external constraints affect the translation of scientific progress into commercial output. Baseline projections indicate a market opportunity exceeding USD 40 billion, with growth largely underpinned by demographic trends and clinical performance rather than short-term macroeconomic conditions. Even in scenarios characterized by geopolitical fragmentation, demand fundamentals remain comparatively stable.
Strategic Implications for the ADC Market Through 2034
Future market leadership is expected to depend on the capability to industrialize ADC platforms efficiently and to demonstrate clear value to regulatory authorities and healthcare payers. As the market matures and competitive intensity increases, execution across development, production, and commercialization is likely to play a more prominent role in shaping competitive positioning than target novelty alone.
About KnowledgeAgent
KnowledgeAgent supports pharmaceutical executives and investors in navigating evolving markets such as ADCs through structured scenario analysis and market modeling. Download the full ADC market forecast to 2034 to explore key takeaways and connect with our Foresight or Life Sciences experts to discuss your market intelligence questions.
Sources: